|
11/22/2023 0 Comments Atomic spectra of potassiumAnd so what you'll often see and you might see something The electron configuration of a sample of a certain type of atom. In this video, we're going to introduce ourselves to the idea of photoelectron spectroscopy. You need more specifics in this question to get a proper answer. And again it's not clear which ones you want to compare. 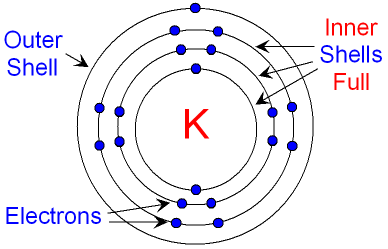
But there are different types of ionization energies we can compare (1st, 2d, 3rd, etc.). Like I've said before binding energy in this instance means ionization energy. So a discussion with a partially filled p subshell with two electrons versus three electrons will require discussion about that stability, and so it's not clear if you simply mean partially filled and specific type of partial filling. But a half-filled subshell creates a certain degree of stability. Having a p subshell partially filled means we could have anywhere from one electron to five electrons in it (since a filled p subshell holds six electrons). Partially filled simply means not completely filled and could change the answer. Or it could mean two neutral atoms of different elements which differ by one atomic number. So there's quite a lot of ambiguity with your question so it's hard to give a single answer.įor example, an two atoms which differ by a single electron could mean two atoms of the same element where at least one is an anion. Knowing how tightly they are being held by the effective nuclear charge of the nucleus of an atom gives us an insight into the reactivity of elements. The reason we care so much about the valance electrons is because they are the electrons that do all the exciting reactions and bonding with other atoms. For example the Zeff of lithium is calculated by using Z = 3 and C = 2, so Zeff = 3 - 2 = +1. Where Zeff is the effective nuclear charge, Z is the atomic number (number of protons), and C is the number of core electrons. The formula for calculating the effective nuclear charge of an element is Zeff = Z - C. This difference between the force of attraction to the nucleus and the force of repulsion to core electrons that valance electrons feel is the effective nuclear charge. The valance electrons still feel a force of attraction to the positive charge of the nucleus but they also have to contend with the negative charges of all the cumulative core electrons below repelling them. The outermost shell of electrons are known as the valance electrons, while all the inner shells of electrons are known as core electrons. 
As you go down the rows (or periods) of the periodic table you add more shells of electrons to atoms. Another thing we have to understand is that electrons orbit the nucleus in layers (or shells). Similar charges repel so any one electron feels a force of repulsion to the other electrons as they orbit. The electrons orbiting the atom's nucleus are of course negative in charge. A related and more helpful concept is known as effective nuclear charge. The number of protons tells us the quantity of positive charge in the nucleus and is therefore known as nuclear charge. Atoms of different elements have different numbers of protons and therefore different degrees of attraction between the nucleus and the electrons. Opposite charges attract each other so the positive protons in the nucleus and the negative electrons in orbit feel a force of attraction between them. Surrounding this nucleus are the electrons (which are negative in charge) who orbit the nucleus. In an atom, the center is known as the nucleus which holds an atom's protons (which are positive in charge) and its neutrons (Which are neutral in charge).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
